A Complete Guide to Oral Collagen Peptides and Hyaluronic Acid Supplements: Bioavailability, Dosage Form Selection, and OEM Challenges (2026 Aidacru In-Depth Analysis)
Date: 2026-04-25 Categories: Blog Hits: 210
The boundaries of "skincare" are being redefined. According to a 2025 report by Mordor Intelligence, the global oral beauty supplement market has exceeded $7 billion and is projected to continue growing at a CAGR of 8.9% by 2030. Within the "Ingestible Beauty" category, oral collagen peptides and hyaluronic acid have risen from niche concepts to mainstream shelf essentials.
However, the stability of active ingredients, bioavailability, and taste are the three core challenges hindering the quality of oral beauty products. This article provides brands with a comprehensive guide to product selection and supply chain management from the perspectives of formulation science and OEM manufacturing processes, and systematically breaks it down based on Aidacru's practical project experience in the development of oral beauty supplement OEM/ODM.
一、Scientific Basis: Why is ingested collagen effective?
The controversy surrounding oral collagen has been long-standing, with the core question being: if proteins are broken down by digestive enzymes, can they truly reach the skin?
The answer lies in molecular weight. A double-blind, randomized controlled trial published in the journal *Nutrients* in 2019 showed that oral administration of collagen hydrolysate with a molecular weight below 3,000 Daltons significantly improved skin elasticity and hydration (Proksch et al., 2019, Nutrients). Small peptides can be directly absorbed into the bloodstream from the intestines in dipeptide and tripeptide forms, promoting type I collagen synthesis in skin fibroblasts.
Oral hyaluronic acid also has well-documented effects. A 2021 study in the journal *Applied Sciences* showed that low molecular weight oral hyaluronic acid (<50 kDa) can be absorbed via the intestinal lymphatic pathway, with effective concentrations detected in joint and skin tissues (Oe et al., 2021).
二、Dosage Form Comparison: Liquid vs. Capsules vs. Gummies, Which Has Higher Bioavailability?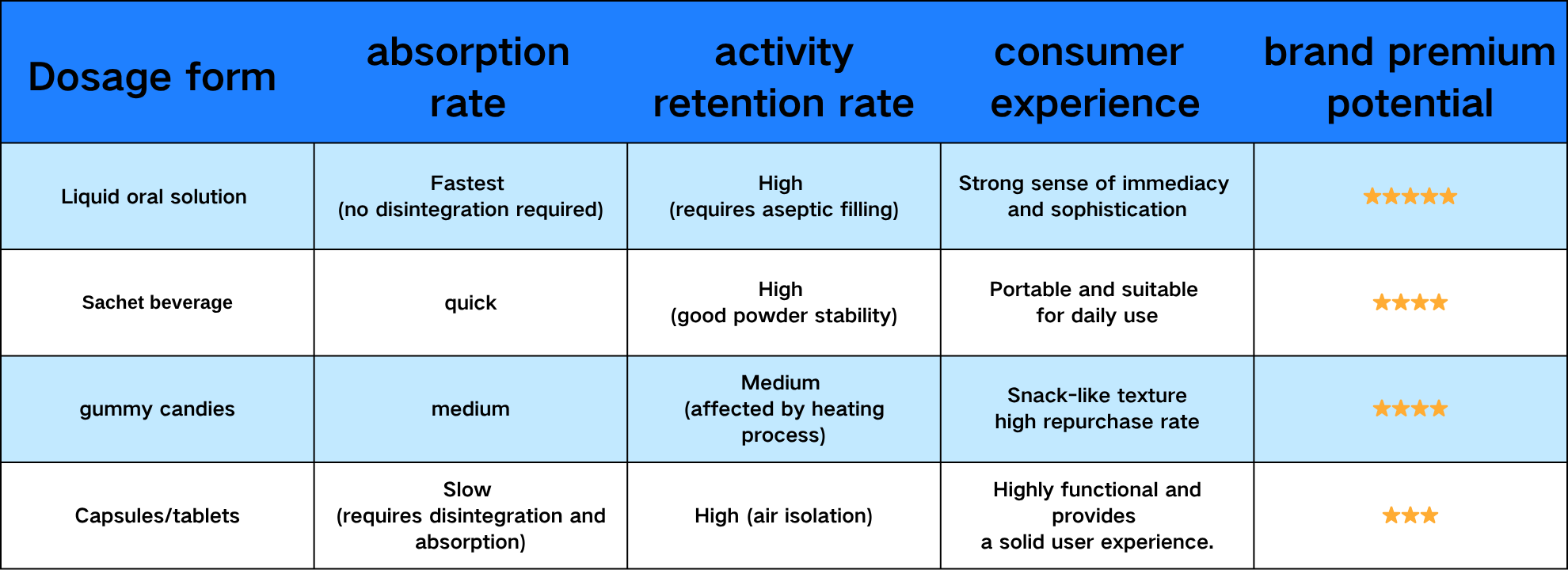
Liquid dosage forms have become the preferred choice for high-end oral beauty brands due to their direct sensory experience—consumers feel the effects the moment they ingest it, giving them a natural advantage in brand premium and social media marketing.
三、 Core Challenges in OEM Manufacturing: The Stability Challenge of Active Ingredients
Hyaluronic Acid's pH Stability
Hyaluronic acid is highly susceptible to degradation in liquid environments due to pH changes. When the solution pH is below 4.0 or above 8.0, the viscoelasticity of hyaluronic acid decreases significantly, leading to a decline in effectiveness within the shelf life.
• Solution from Professional Factories: Precisely adjusting the acid-base buffer system to maintain the pH of liquid products within the optimal range (typically 5.5-7.0). In actual OEM projects, Aidacru typically uses multi-level buffer systems and long-term stability testing to ensure that hyaluronic acid maintains its activity and rheological properties throughout its shelf life.
• Verification Method: Requires the factory to provide a 6-month accelerated stability test report (40℃/75%RH conditions).
Odor Masking Issues of Collagen Peptides
Collagen peptides derived from fish or poultry often carry a fishy or gamey odor, which is the leading reason for returns of oral beauty products. In Aidacru's past liquid and gummy projects, the flavor system was usually optimized as a separate R&D module to avoid the problem of products that are "effective but difficult to swallow."
• A composite flavor masking solution using plant acids (such as citric acid) + natural fruit flavors
• Customized flavors such as "peach + oolong tea" and "lychee + rose" tailored to Asian consumer preferences
• Requires blind flavor testing during the sampling stage (usually requiring 10-20 consumers to participate).
四、Core Challenges of OEM Manufacturing, Part Two: Aseptic Production and Global Compliance
Liquid oral beauty products require near-pharmaceutical-level microbial control. Without chemical preservatives (for the clean label market), aseptic filling is the only safe solution.
• Key Hardware: Class 100,000 (ISO Class 8) or Class 10,000 (ISO Class 7) cleanroom for filling.
• Required Certifications: WHO-GMP, FDA facility registration, and compliance with EC 1223/2009 standards for the EU market.
• Raw Material Traceability: Requires certificates of origin and third-party inspection reports for core raw materials (collagen peptides, hyaluronic acid).
Aidacru's factory completes fully enclosed aseptic filling in a Class 100,000 cleanroom, fully complying with WHO-GMP and FDA registration standards. It insists on directly sourcing core raw materials from top-tier production areas such as France and Germany, with complete traceability documentation for each batch.
五、Brand Solution Example: The Complete Path from Zero to One for Liquid Collagen Products
In Aidacru's past projects, a German skincare brand client sought to expand its oral product line, facing three challenges: the fishy smell of liquid collagen, production qualification audits, and initial inventory risks.
The solution path was as follows:
1. Flavor Customization Stage (2 weeks): Conducting consumer flavor preference research targeting the target audience (urban women aged 25-40) and customizing a natural fruit flavor masking solution.
2. Qualification Preparation Stage (Simultaneous): The factory provided complete ISO certificates and international quality control reports to support compliance audits on independent e-commerce platforms.
3. Small-Batch Testing (4-6 weeks): Initial production with a low MOQ, launching the product on social media to test market reaction.
4. Mass Production After Data Validation: First-month repurchase rate data supported formal mass production scheduling.
Results: The product successfully increased its price by 20%, and the monthly active repurchase rate remained stable at over 28%, completing the transition from "OEM product" to "brand asset."
⚡ Frequently Asked Questions (FAQ)
Q: What molecular weight of oral collagen is considered "effective"?
A: According to existing clinical studies (Proksch et al., Nutrients 2019), collagen hydrolysates with a molecular weight below 3,000 Daltons have the best intestinal absorption rate and skin efficacy evidence. Suppliers should be requested to provide raw material molecular weight distribution test reports during procurement.
Q: Is it really possible to produce liquid collagen products without preservatives?
A: Yes, but the production environment requirements are extremely high. In actual projects, Aidacru typically achieves a stable long shelf life through a combination of aseptic filling systems and physical oxygen barrier technologies. Aseptic filling processes (cleanliness level of 100,000 or higher) are required, combined with nitrogen filling, aluminum foil sealing, and other physical oxygen barrier methods, typically achieving a shelf life of 18-24 months.
Q: What compliance documents are required for producing oral beauty products?
A: Entering the US market requires FDA facility registration (cGMP compliance) and product labeling in accordance with 21 CFR Part 101; entering the EU market requires compliance with the Food Supplements Directive 2002/46/EC; some e-commerce platforms (such as Amazon) also require a third-party Certificate of Accreditation (COA) and a Heavy Metal Test Report.
Q: Which is more suitable for startup brands: collagen gummies or collagen liquid oral liquid?
A: Both have their advantages: gummies offer a better user experience and higher repurchase rates, but the production process is more complex; liquid oral liquids offer greater brand premium potential, but have stricter requirements for aseptic filling equipment. Startups with limited funds are advised to start with powdered gummies to reduce MOQ risk, and then upgrade to liquid formulations after market validation.
For details on our formulation solutions and agency qualifications for oral beauty products, please contact the Aidacru R&D team for a specialized consultation.
