2026 Functional Gummy OEM Manufacturer Selection Guide: 5 Pitfalls to Avoid (Including MOQ and Process Comparison)
Date: 2026-04-25 Categories: Blog Hits: 225
If there's one dosage form that will be as addictive as snacks for consumers in 2026, supplement gummies are undoubtedly the answer. According to a 2025 report by Grand View Research, the global supplement gummy market is projected to exceed $35 billion by 2030, with a compound annual growth rate (CAGR) of 12.3%. Gummies have been the preferred dosage form for supplement brands launching their products for four consecutive years.
However, the barriers to entry for gummy candy OEM manufacturing are far higher than imagined. Many brands often encounter disastrous problems after signing contracts, such as melting and sticking together, insufficient active ingredient content, and a rubbery texture. This guide, summarized by our Aidacru team of production experts, covers five core dimensions of the entire gummy candy selection process.
一、Temperature and Humidity Control: The First Lifeline of Contract Factories
Melting and sticking together is the most common disaster scenario for gummies, stemming from a lack of sophisticated environmental management at the contract manufacturer. When entire batches of products arrive at US/European warehouses and clump together, it directly leads to the return of the entire shipment, resulting in losses of up to hundreds of thousands of dollars.
How to assess a factory's temperature and humidity management capabilities?
• We require the factory to provide the hardware parameters of an independent temperature and humidity controlled packaging room (temperature ±1℃, humidity ±5% RH).
• We inquire whether 24-hour dynamic environmental data recording is implemented (historical data samples are required).
• We require a high-temperature stability test report (typically 40℃/75% RH, 6-month accelerated testing).
All gummy candy production and packaging processes at our Aidacru factory are completed in a Class 100,000 cleanroom with constant temperature and humidity (ISO Class 8 standard), with 24-hour continuous dynamic environmental monitoring. Each batch of products must undergo rigorous accelerated stability testing before leaving the factory to ensure that the product maintains its intact appearance under simulated high-temperature sea transport conditions.
二、Active Ingredient Retention Rate: The Bottom Line for Functionality
Gummy production requires heating to high temperatures, but core ingredients such as Vitamin C, probiotics, and plant extracts are extremely heat-sensitive. Some small factories, due to outdated processes, may only have 60%-70% of the claimed core ingredient content in their finished products. This is not only product fraud but also a trigger for regulatory risks.
According to data from the US FDA, supplement labeling compliance is one of the top three sources of consumer complaints (FDA, Dietary Supplement Warning Letters, 2024).
Process Verification Checklist:
• Confirm whether a low-temperature molding (starchless molding/low-Temperature Depositing) process is available.
• Whether microencapsulation protection is provided for heat-sensitive ingredients.
• Request a recent batch's full-item quality control (FQC/COA) report for active ingredients.
We use an advanced powderless low-temperature molding process to cast active ingredients at the minimum necessary temperature and provide independent microencapsulation treatment for extremely heat-sensitive ingredients such as probiotics. Our in-house laboratory conducts full quality control (FQC) on each batch of ingredients, and a Certificate of Account (COA) is provided with the shipment.
三、Taste and Formula Development Capabilities: The Core of Consumer Retention
Sugar-free gummies are one of the fastest-growing market segments, but artificial sweeteners often result in a rubbery texture or a bitter aftertaste, a key factor affecting repurchase rates.
A 2024 survey of 1,200 US consumers by an independent research firm showed that poor taste was the primary reason for stopping purchases of health supplement gummies (38%).
Evaluation Dimensions:
• Does the factory have pectin formulation capabilities (for the vegan and clean label markets)?
• Size of the flavor customization library (the more mature fruit flavor formulas, the shorter the sampling cycle)
• Does it offer no artificial colors solutions?
Our Aidacru R&D center has a reserve of over 500 mature fruit flavor formulas, specializing in using pectin instead of gelatin to create a chewy, natural texture. Plant-based, clean label, and no artificial color solutions are all mature and in mass production, meeting the labeling requirements of independent websites such as Amazon, TikTokShop, Shopify, and the EU market.
四、 Aidacru's MOQ and Flexible Supply Chain: The Survival Logic of Startup Brands
Too low an MOQ is often a trap—small factories may cut corners on quality control to reduce production costs; too high an MOQ puts startup brands under inventory pressure.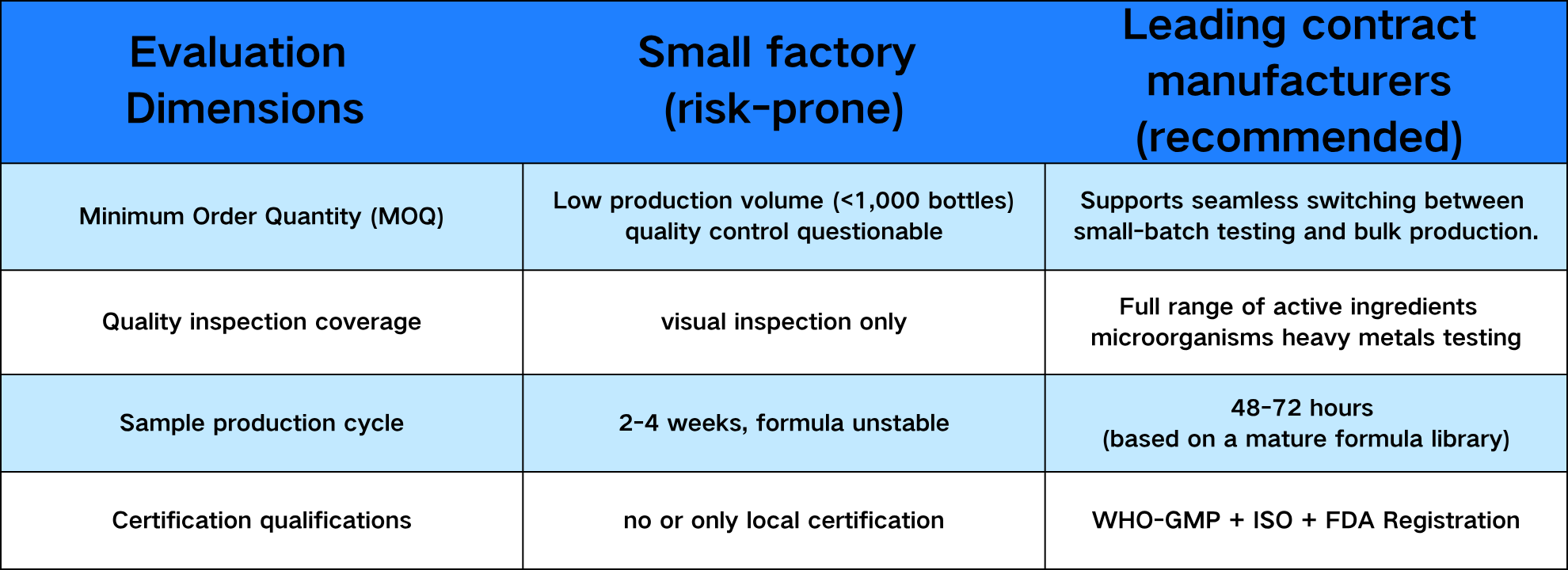
As a leading global manufacturer, Aidacru offers flexible scheduling from small-batch market testing to large-scale production, and supports a one-stop turnkey service from formulation development to packaging design, allowing brands to focus on market operations.
五、Compliance Certifications: The Passport to International Markets
For entry into the US market, cGMP (Current Good Manufacturing Practice) certification is a basic requirement of the FDA; for entry into the EU, compliance with EN ISO 22000 food safety management standards is required. Some contract manufacturers only hold domestic certifications, which may lead to problems during customs clearance.
• Mandatory Certifications: WHO-GMP, FDA Facility Registration Number (FEI), ISO 9001/22000
• Requires third-party audit reports within the last 3 years
• Confirmation that the factory can provide a product compliance documentation package that meets the requirements of the target market (US/EU/Australia)
⚡ Frequently Asked Questions (FAQ)
Q: What is the minimum order quantity (MOQ) for functional gummy manufacturing?
A: The industry standard MOQ is typically between 5,000-10,000 bottles, depending on the complexity of the formula and packaging. Aidacru, a leading manufacturer with flexible production capacity, supports market test orders with lower MOQs, suitable for DTC startups to verify market response before mass production.
Q: How are the taste issues of sugar-free gummies addressed?
A: The key lies in the combination of sugar substitutes and the choice of colloid. Professional factories customize the sugar substitute ratio according to the target audience (e.g., ketogenic diet, children, elderly) and improve elasticity and chewiness through a pectin + carrageenan composite colloid formula, avoiding a rubbery feel and bitter aftertaste.
Q: How is the live probiotic count in gummies guaranteed to meet standards even after expiration?
A: The core lies in two points: first, selecting heat- and acid-resistant bacterial strains (such as Bacillus coagulans); second, protecting live bacteria during the production process through microencapsulation technology. Aidacru's factory adheres to the principle of adding more live bacteria during product design, ensuring that the live bacteria count at expiration is not lower than the claimed value on the label.
Q: How long does it take from sampling to the first batch of bulk orders?
A: Based on a mature formula, sampling typically takes 3-7 business days; after sample confirmation, the first batch of bulk production takes approximately 4-6 weeks (including testing and packaging). If a completely new formula is involved, the overall cycle extends to 8-12 weeks.
Q: Does the gummy candy OEM manufacturer need FDA registration?
A: Yes. For exporting supplement gummies to the US market, the production facility must be registered with the FDA and comply with 21 CFR Part 111 cGMP regulations.
For Aidacru's gummy candy sampling plan or factory qualification documents, please contact our technical advisory team for one-on-one consultation.
